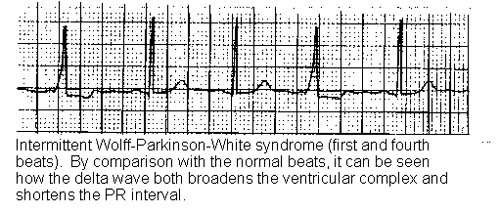
- от автора Jose
Wolff-Parkinson-White Syndrome
Dr Colin Tidy, Reviewed by Dr John Cox | Last edited 16 Feb 2015 | Certified by The Information Standard
Professional Reference articles are designed for health professionals to use. They are written by UK doctors and based on research evidence, UK and European Guidelines. You may find the Wolff-Parkinson-White Syndrome article more useful, or one of our other health articles.
Wolff-Parkinson-White Syndrome
In this article
Wolff-Parkinson-White (WPW) syndrome is the most common of the ventricular pre-excitation syndromes. Others include Lown-Ganong-Levine syndrome and Mahaim-type pre-excitation. They are important because of the association with paroxysmal tachycardias that can result in serious cardiovascular complications and sudden death. In ECG terms they are important to recognise, because of the risk of misdiagnosis. WPW syndrome was first described by Drs Wolff, Parkinson, and White in 1930. [1]
WPW syndrome is a congenital abnormality which can result in supraventricular tachycardia (SVT) that uses an atrioventricular (AV) accessory tract. The accessory pathway may also allow conduction during other supraventricular arrhythmias, such as atrial fibrillation or flutter. The majority of patients with ECG findings of pre-excitation do not develop tachyarrhythmias. WPW syndrome is classified into two types according to the ECG findings:
- Type A: the delta wave and QRS complex are predominantly upright in the precordial leads. The dominant R wave in lead V1 may be misinterpreted as right bundle branch block.
- Type B: the delta wave and QRS complex are predominantly negative in leads V1 and V2 and positive in the other precordial leads, resembling left bundle branch block.
Causes
- An accessory pathway is likely to be congenital, although it presents in later years and may appear to be acquired.
- May be associated with congenital cardiac defects, Ebstein’s anomaly, mitral valve prolapse, hypertrophic cardiomyopathy or other cardiomyopathies.
Ep >[2]
Presentation
- SVT in WPW syndrome may begin in childhood or not appear clinically until m >[4]
- Symptomatic: palpitations, light-headedness or syncope.
- The tachycardia that produces symptoms may be an SVT, atrial fibrillation, or atrial flutter.
- Paroxysmal SVT can be followed after termination by polyuria, which is due to atrial dilatation and release of atrial natriuretic factor.
- Sudden death: from deterioration of pre-excited atrial fibrillation into ventricular fibrillation. [5]
- During SVT, the rhythm is constant and regular, with constant intensity of the first heart sound.
- The jugular venous pressure may be elevated but the waveform remains constant.
- Clinical features of associated cardiac defects may be present — eg, mitral valve prolapse, cardiomyopathy.
Differential diagnosis [6]
- Atrial fibrillation.
- Atrial flutter.
- Atrioventricular nodal re-entry tachycardia (AVNRT).
- Sinus node dysfunction.
- Ventricular fibrillation.
- Ventricular tachycardia.
- Ebstein’s anomaly.
- Lown-Ganong-Levine syndrome.
- Other causes of syncope.
Investigations
ECG [7]

- The electrical impulse may travel at the same speed as along the normal system (bundle of His), with no pre-excitation and the ECG is normal. The condition is described as latent, until the rate exceeds the refractory period of the AV node.
- >[8, 9]
- Repolarisation abnormalities are common in patients with WPW syndrome.
- AV re-entry tachycardia, or circus movement tachycardia:
- The accessory pathway only conducts in a retrograde manner and the condition remains latent until triggered.
- A premature atrial extrasystole, finding the accessory pathway refractory, travels via the AV node, with reactivation back from the ventricles along the accessory pathway (retrograde conduction), if time has allowed recovery of excitability.
- Therefore, a circuit is established, being seen as a tachycardia, with normal QRS but with inverted P waves (because of retrograde atrial activation).
- Pre-excited atrial fibrillation:
- Conduction is rap >
Other investigations
- A variety of recording devices can be used if arrhythmias are infrequent, including 24-hour Holter monitors, event monitors and implantable loop recorders.
- Stress testing may help to diagnose transient paroxysmal dysrhythmia, determine the relationship between exercise and tachycardia or evaluate the effect of medication.
- Routine blood tests may be needed to help rule out non-cardiac conditions triggering tachycardia — eg, FBC, U&E and creatinine, LFTs, TFTs and blood levels of anti-arrhythmic drugs.
- Echocardiogram: may be needed to assess left ventricular function and wall motion and to help rule out valvular disease, Ebstein’s anomaly, hypertrophic cardiomyopathy (in which the incidence of accessory pathways is increased), or other congenital cardiac defects.
- Intracardiac or oesophageal electrophysiological studies may be useful in identifying accessory pathways and during surgery to map areas that require ablation.
Management
Asymptomatic patients may just need periodic review. The main forms of treatment are drug therapy, radiofrequency (RF) ablation and surgical ablation. Ablation is the first-line treatment for symptomatic WPW syndrome. It has replaced surgical treatment and most drug treatments. [10] Drug treatment may still be appropriate for patients who refuse ablation or as an interim control measure if they are at high risk of complications.
- Patients who present with tachyarrhythmic symptoms require drug therapy to prevent further episodes. Drugs used include amiodarone and sotalol. [11]
- A membrane-active anti-arrhythmic drug ( >
Rare Disease Database
Wolff Parkinson White Syndrome
Synonyms of Wolff Parkinson White Syndrome
- Accessory Atrioventricular Pathways
- Preexcitation Syndrome
- WPW Syndrome
General Discussion
Wolff-Parkinson-White (WPW) syndrome is a rare congenital heart disorder involving irregularities in the electrical system of the heart. In individuals with WPW syndrome, an abnormal alternate electrical pathway (accessory pathway), exists between the atrium and the ventricle, resulting in abnormal heartbeat rhythms (arrhythmias) and faster than normal heartbeats (tachycardia).
The normal heart has four chambers. The two upper chambers are the atria and the two lower chambers are the ventricles. Within the right atrium of a normal heart is a natural pacemaker that initiates and controls the heartbeat. The electrical stimulus travels from the pacemaker (sinoatrial or SA node) to the ventricles along a specific pathway consisting of conducting tissue and known as the AV (atrioventricular) node. The extra electrical pathway in individuals with WPW syndrome bypasses the normal route and causes the ventricles to beat earlier than normal (preexcitation) and can allow electrical impulses to be conducted in both directions (i.e., from the atria to the ventricles and from the ventricles to the atria).
Signs & Symptoms
The symptoms associated with WPW syndrome vary greatly from case to case. Some individuals may not have any abnormal heartbeats or associated symptoms (asymptomatic). Although the disorder is present at birth (congenital), symptoms may not become apparent until adolescent or early adulthood.
Individuals with WPW syndrome may exhibit one or more irregular heartbeats especially episodes of abnormally fast heartbeats that originate above the ventricles (supraventricular tachycardias). These episodes often begin and stop abruptly and may last a few minutes to a few hours. The frequency of episodes varies from case to case. Some individuals may experience episodes every week, others may only have a few sporadic episodes.
A variety of symptoms may occur during these episodes including palpitations, difficulty breathing (dyspnea), light-headedness, chest pain, decreased exercise tolerance, anxiety, and dizziness. In some cases, affected individuals may lose consciousness (syncope).
Some individuals with WPW syndrome may experience an atrial flutter, in which the atrium beats regularly at an extremely rapid rate or atrial fibrillation, in which there is rapid, irregular twitching of the muscular wall. In extremely rare cases, affected individuals may develop ventricular fibrillation, a serious condition in which in which the heart’s normal electrical activity becomes disordered resulting in uncoordinated heartbeats and malfunction of the main pumping chambers of the heart (ventricles). Although rare in WPW syndrome, ventricular fibrillation can potentially result in cardiac arrest and sudden death.
Causes
Most cases of WPW syndrome occur randomly in the general population for no apparent reason (sporadically) and do not run in families. Some cases of WPW syndrome run in families and may be inherited as an autosomal dominant trait.
No specific genetic mutation has been identified in individuals with isolated WPW syndrome and the exact role genetics plays in the development of WPW syndrome is not fully understood. However, a rare autosomal dominant disorder known as familial Wolff-Parkinson-White syndrome has been linked to chromosome 7. Investigators have determined that mutations of the gamma-2 regulatory subunit of AMP-activated protein kinase (PRKAG2) gene located on the long arm (q) of chromosome 7 (7q36) cause this disorder, which comprises the features of WPW syndrome, progressive conduction block, and overgrowth of a portion of the heart (cardiac hypertrophy).
Some researchers believe that familial WPW syndrome is a glycogen storage disorder, a group of disorders in which stored glycogen, which is normally broken down (metabolized) into glucose to supply energy, accumulates in various organs of the body. WPW syndrome is known to occur as part of other glycogen storage disorders, specifically Pompe disease or Danon disease.
Approximately 7 to 20 percent of individuals with WPW syndrome have a congenital heart defect, such as Ebstein’s anomaly, a condition in which the tricuspid valve is malformed. The tricuspid valve connects the right atrium to the right ventricle.
The symptoms of WPW syndrome result from the presence of an alternate electrical pathway. The normal heart has one conduction pathway (the Bundle of His) along which electrical impulses are transmitted from the small chambers of the heart (the atria) to the large chambers (the ventricles). These electrical impulses induce the muscles of the atria and then the ventricles, to contract and relax, pumping blood throughout the body. Individuals with WPW syndrome have a second abnormal conduction pathway, the Bundle of Kent, which sends extra electrical impulses from the muscles of the atria to those of the ventricles. These extra electrical impulses bypass the normal route and disrupt the normal rhythm of heartbeats and generate irregularities, usually exceptionally rapid heartbeats known as “atrial flutter, atrial fibrillation, or paroxysmal supraventricular tachycardia.” The exact cause of alternate pathways is unknown.
Affected Populations
WPW syndrome is often present at birth (congenital), but may not be detected until adolescence or later. Peak incidence has been reported in individuals between 30 and 40 years old in otherwise healthy adults. Some reports suggest that WPW syndrome occurs in males more often than females. The disorder’s estimated prevalence is .1-3.1 per 1,000 people in the United States.
Related Disorders
Symptoms of the following disorders can be similar to those of WPW syndrome. Comparisons may be useful for a differential diagnosis.
Lown-Ganong-Levine (LGL) syndrome is a rare congenital heart disorder involving irregularities in the electrical system of the heart. The ventricles receive part or all of their electrical impulses from an irregular conduction pathway (alternate pathway) instead of from the Bundle of His. Individuals with LGL syndrome may experience a variety of irregular heartbeats including atrial flutter, atrial fibrillation and paroxysmal atria arrhythmias. Symptoms associated with these irregular heartbeats include faintness, weakness, palpitations and nausea. The specific location of the alternate pathway in LGL syndrome is not known.
Sick sinus syndrome is a rare heart (cardiac) disorder characterized by irregular heart rhythm (arrhythmias). Affected individuals may experience excessively slow heartbeats (bradycardia) and rapid heartbeats (tachycardia). Additional heart rhythm abnormalities may occur including gradual supraventricular tachycardia, atrial flutter, and atrial fibrillation. Palpitations, weakness, faintness, and nausea are common symptoms. Most cases of sick sinus syndrome occur in individuals 50 years old or older. Sick sinus syndrome is caused by malfunction of the heart’s natural pacemaker (sinoatrial node).
The abnormal heart rhythms associated with WPW syndrome (e.g., atrial flutter, supraventricular tachycardia) may occur as isolated primary findings or secondary to structural heart disease. Such alternate causes of abnormal heart rhythms should be differentiated from WPW syndrome.
Diagnosis
A diagnosis of WPW syndrome is suspected based upon a thorough clinical evaluation, a detailed patient history and a variety of specialized tests. Such tests might include an electrocardiogram (ECG), Holter monitor, and electrophysiology study. An electrocardiogram records the heart’s electrical impulses and may reveal abnormal electrical patterns. A Holter monitor is a portable device designed to continuously monitor the electrical activity of the heart. A Holter monitor is usually worn for 24 hours. During electrophysiology study, a thin tube (catheter) is inserted into a blood vessel and threaded to the heart where it measures electrical activity. Each of these specialized tests can detect abnormal heart rhythms associated with WPW syndrome.
Some individuals with WPW syndrome may be clinically «silent» meaning that they have no symptoms associated with the disorder including no abnormal findings on various heart tests.
Standard Therapies
Treatment
The treatment of Wolff-Parkinson-White syndrome may include observation without specific intervention, the use of various medications, and a surgical procedure known as catheter (radiofrequency) ablation.
Specific therapeutic procedures and interventions may vary, depending upon numerous factors, such as the type of arrhythmia present; the frequency; the type and severity of associated symptoms; risk of cardiac arrest; an individual’s age and general health; and/or other elements. Decisions concerning the use of particular interventions should be made by physicians and other members of the health care team in careful consultation with the patient, based upon the specifics of his or her case; a thorough discussion of the potential benefits and risks; patient preference; and other appropriate factors.
In some individuals without symptoms (asymptomatic cases) no therapy may be necessary. Regular, follow-up visits are necessary to monitor heart function.
Various medications can be used to control episodes of arrhythmias in some individuals with WPW syndrome. Such medications, known as antiarrhythmics, include adenosine, procainamide, sotalol, flecainide, ibutilide, and amiodarone. Calcium channel blockers such as verapamil may also be used. Certain medications such as verapamil may increase the risk of ventricular fibrillation and should be used with caution.
The heart drug, digoxin, is contraindicated in adults with WPW syndrome. However, it is sometimes used for prophylaxis treatment of infants with WPW syndrome who do not have ventricular preexcitation.
In some cases, medications may not be sufficient to control episodes of abnormal heartbeats or individuals may not be able to tolerate the medicine. A surgical procedure known as catheter ablation may be used in such cases. This procedure may also be used in individuals who are at high risk of developing cardiac arrest and sudden death including some asymptomatic individuals.
During a catheter ablation, a small thin tube (catheter) is inserted into the heart and guided to the abnormal pathway where high-frequency electrical energy is used to destroy (ablate) the tissue making up the abnormal pathway. This form of therapy has an extremely high success rate and may end the need of medications in many individuals.
In the past, open heart surgery had been used to treat individuals with WPW syndrome. Because of the success of the less invasive procedure, catheter (radiofrequency) ablation, open heart surgery is rarely performed for individuals with WPW syndrome.
Investigational Therapies
Information on current clinical trials is posted on the Internet at www.clinicaltrials.gov. All studies receiving U.S. government funding, and some supported by private industry, are posted on this government web site.
For information about clinical trials being conducted at the NIH Clinical Center in Bethesda, MD, contact the NIH Patient Recruitment Office:
Tollfree: (800) 411-1222
TTY: (866) 411-1010
For information about clinical trials sponsored by private sources, contact:
Supporting Organizations
- American Heart Association
- 7272 Greenville Avenue
- Dallas, TX 75231
- Phone: (214) 784-7212
- Toll-free: (800) 242-8721
- Email: Review.personal.info@heart.org
- Website: http://www.heart.org
- Cardiomyopathy Association
- Chiltern Court, Unit 10
- Asheridge Rd Ind
- Chesham, HP5 2PX United Kingdom
- Phone: 4401494791224
- Toll-free: (800) 018-1024
- Email: info@cardiomyopathy.org
- Website: http://www.cardiomyopathy.org
- Genetic and Rare Diseases (GARD) Information Center
- PO Box 8126
- Gaithersburg, MD 20898-8126
- Phone: (301) 251-4925
- Toll-free: (888) 205-2311
- Website: http://rarediseases.info.nih.gov/GARD/
- NIH/National Heart, Lung and Blood Institute
- P.O. Box 30105
- Bethesda, MD 20892-0105
- Phone: (301) 592-8573
- Email: nhlbiinfo@rover.nhlbi.nih.gov
- Website: http://www.nhlbi.nih.gov/
References
Behrman RE, Kliegman RM, Jenson HB. Eds. Nelson Textbook of Pediatrics. 17th ed. Elsevier Saunders. Philadelphia, PA; 2005:1559-61.
Kasper, DL, Fauci AS, Longo DL, et al. Eds. Harrison’s Principles of Internal Medicine. 16th ed. McGraw-Hill Companies. New York, NY; 2005:1350-1.
Foster V, Alexander RW, O’Rourke RA, et al. Eds. Hurst’s The Heart. 11thed. McGraw-Hill Companies. New York, NY; 2004:855-870.
Light PE. Familial Wolff-Parkinson-White syndrome: a disease of glycogen storage or ion channel dysfunction? J Cardiovasc Electrophysiol. 2006;17:S158-61.
Ehtisham J, Watkins H. Is Wolff-Parkinson-White syndrome a genetic disease? J Cardiovasc Electrophysiol. 2005;16:1258-62.
Sidhu JS, Rajawat YS, Tapan GR, et al., Transgenic mouse model of ventricular preexcitation and atrioventricular reentrant tachycardia induced by an AMP-activated protein kinase loss-of-function mutation responsible for Wolff-Parkinson-White syndrome. Circulation. 2005;111:21-9.
Redfearn DP, Krahn AD, Skanes AC, Yee R, Klein GJ. Use of medications in Wolff-Parkinson-White syndrome. Expert Opin Pharmacother. 2005;6:955-63.
Huang D, Yamauchi K, Inden Y, et al., Use of artificial neural network to localize accessory pathways of Wolff-Parkinson-White syndrome with 12-lead electrocardiogram. Med Inform Internet Med. 2005;30:277-86.
Pappone C, Manguso F, Santinelli R, et al., Radiofrequency ablation in children with asymptomatic Wolff-Parkinson-White syndrome. N Engl J Med. 2004;351:1197-205.
Milliez P, Slama R. Wolff-Parkinson-White syndrome. Rev Pract. 2004;54:1747-53.
Arad M, Benson D, Perez-Atayde AR, et al., Constituively activate AMP kinase mutations cause glycogen storage disease mimicking hypertrophic cardiomyopathy. J Clin Invest. 2002;109:357-62.
Gollob MH, Green MS, Tang MSL, et al., Identification of a gene responsible for familial Wolff-Parkinson-White syndrome. N Engl J Med. 2001;344:1823-31.
MacRae CA, Ghaisas N, Kass S, et al., Familial hypertrophic cardiomyopathy with Wolff-Parkinson-White syndrome maps to a locus on chromosome 7q3. J Clin Invest. 1995;96:1216-20.
FROM THE INTERNET
The Cleveland Clinic. Wolff-Parkinson-White syndrome (WPW). April 2001. Available at: http://www.clevelandclinic.org/heartcenter/pub/guide/disease/electric/wpw.htm Accessed On: June 29, 2006.
Hemingway TJ. Wolff-Parkinson-White syndrome. Emedicine Journal, June 14 2006. Available at: http://www.emedicine.com/emerg/topic644.htm Accessed on: June 29, 2006.
Singh VN. Wolff-Parkinson-White syndrome. Emedicine Journal, October 28, 2004. Available at: http://www.emedicine.com/med/topic2417.htm Accessed on: June 29, 2006.
Bouvagnet P. Wolff-Parkinson-White syndrome. Orphanet encyclopedia, March 2002. Available at: http://www.orpha.net/consor/cgi-bin/OC_Exp.php?Lng=GB&Expert=907 Accessed on: April 4, 2006.
McKusick VA., ed. Online Mendelian Inheritance in Man (OMIM). Baltimore. MD: The Johns Hopkins University; Entry No:194200; Last Update:10/08/2004. Available at: http://www.ncbi.nlm.nih.gov/entrez/dispomim.cgi? >
Years Published
The information in NORD’s Rare Disease Database is for educational purposes only and is not intended to replace the advice of a physician or other qualified medical professional.
National Organization for Rare Disorders (NORD)
55 Kenosia Ave., Danbury CT 06810 • (203)744-0100Синдром Вольфа-Паркинсона-Уайта (синдром WPW)
Тел.: 8-800-25-03-03-2
(бесплатно для звонков из регионов России)
Санкт-Петербург, наб. реки Фонтанки, д. 154
Тел.: +7 (812) 676-25-25Санкт-Петербург, В.О., Кадетская линия, д. 13-15
Тел.: +7 (812) 676-25-25Санкт-Петербург, ул. Циолковского, д.3
Тел.: +7 (812) 676-25-10Синдром WPW характеризуется наличием приступов пароксизмальной тахикардии по механизму «re-entry» (повторный вход волны возбуждения) по дополнительному (врожденному) атриовентрикулярному соединению. На ЭКГ у таких пациентов, как правило, есть характерные изменения: наличие дельта — волны, расширенный QRS — комплекс и укороченный интервал PQ (PR).
Именно наличие приступов тахикардии при характерных изменениях на ЭКГ отличает синдром WPW от одноименного феномена. Врожденный дополнительный путь проведения (пучок Кента) проводит импульсы возбуждения от предсердий к желудочкам в обход обычному пути (через атриовентрикулярный-АВ-узел), что приводит к раннему возбуждению желудочков, а при определенных условиях — развитию тахикардии. Среди общего количества пациентов с этой патологией, при наличии признаков предвозбуждения по ЭКГ, у 40-65% пациентов отмечаются приступы тахикардий. У 30% пациентов с синдромом WPW встречается фибрилляция предсердий (ФП). Развитие пароксизма ФП с проведением по дополнительному пути может привести к чрезвычайно быстрой тахикардии с высокой ЧСС, что представляет угрозу для жизни.

Помимо описанного дополнительного пути проведения, также существуют другие врожденные дополнительные пути проведения: волокна Махейма, тракт Джеймса, тракт Брашенмаше. Анатомически эти пути могут соединять: АВ-узел и межжелудочковую перегородку, правое предсердие и ствол пучка Гиса, пучок Гиса и миокард желудочков. Наличие у пациентов врожденных дополнительных путей проведения импульса возбуждения, приступов тахикардии и пароксизмов ФП, определяют необходимость проведения электрофизиологического исследования сердца и катетерной аблации (деструкции) этих путей.
Основные проявления синдрома WPW
- Внезапно возникающий приступ сердцебиения.
- Перебои в работе сердца.
- Пульсация в голове или горле.
- Общая слабость, головокружение, снижение толерантности к физической нагрузке, повышенная утомляемость на фоне приступа.
- Возможна потеря сознания.
- Одышка.
- На фоне приступа возможно развитие головокружения или потери сознания.
- Снижение артериального давления (артериальная гипотензия или нестабильное АД).
Диагностика синдрома WPW
Врач поликлиники, «скорой помощи» или кардиолог собирают анамнез, проводят физикальное обследование, и различные инструментально-диагностические методы обычно после приступа. Выполняется ЭКГ. Наиболее важным является регистрация приступа тахиаритмии на ЭКГ (для предъявления аритмологу). Сегодня пациентам доступна консультация врача кардиохирурга-аритмолога в специализированных или многопрофильных клиниках, который может определить необходимость и возможность эндоваскулярного интервенционного (малотравматичного) лечения.
В Клинике есть все возможные способы диагностики и лечения тахикардий. Основные из них:
1. ЭКГ в 12 отведениях.
2. Суточное мониторирование ЭКГ.
3. Эндокардиальное электрофизиологическое исследование сердца (эндо-ЭФИ) – чреспищеводное (проводят в амбулаторных условиях) и эндокардиальное (проводят в стационаре).
Эндокардиальное ЭФИ сердца осуществляется в рентген-операционной. Этот метод позволяет произвести оценку функционального состояния проводящей системы сердца и выяснить механизм возникновения аритмии, определить показания к проведению радиочастотной катетерной аблации (РЧА) дополнительных путей проведения и аритмогенных зон. То есть, диагностическая процедура может перейти в лечебную.
Лечение синдрома WPW
Выбор способа лечения пациента осуществляется специалистом с учетом клинической картины заболевания, данных инструментально-диагностических исследований и рекомендаций Всероссийского научного общества аритмологов, Всероссийского научного общества кардиологов.
Самостоятельный прием препаратов, самолечение различными методами является крайне не желательным и не безопасным с учетом неизвестного характера, механизма и причин тахикардий.
Существует несколько способов лечения синдрома WPW:
1. Антиаритмическая терапия — при постоянном приеме медикаментозных препаратов.
2. Электрофизиологические методы:
- кардиоверсия/дефибрилляция (синхронизированная с ЭКГ наружная дефибрилляция);
- катетерная аблация дополнительного пути проведения.
Катетерная аблация («прижигание») дополнительного пути проведения является высокоэффективным и радикальным методом лечения синдрома WPW. Операция, в среднем, выполняется в течение 1 часа, и через сутки пациент может быть выписан из стационара.
На базе кабинета рентгенэндоваскулярной диагностики и лечения Клиники высоких медицинских технологий им. Н. И. Пирогова создана ЭФИ-лаборатория, оснащенная самым современным электрофизиологическим оборудованием, что позволяет проводить диагностику и интервенционное лечение любых видов тахиаритмий. На отделении проводятся операции в рамках государственной программы по оказанию высокотехнологичной медицинской помощи (ВМП). Данный вид медицинской помощи осуществляется по квотам, выделяемым Минздравом России. Также пациенты могут быть прооперированы и на хозрасчетной основе с учетом утвержденной в Центре стоимости.
Записаться на консультацию вы можете по тел. 676-25-25
- Conduction is rap >